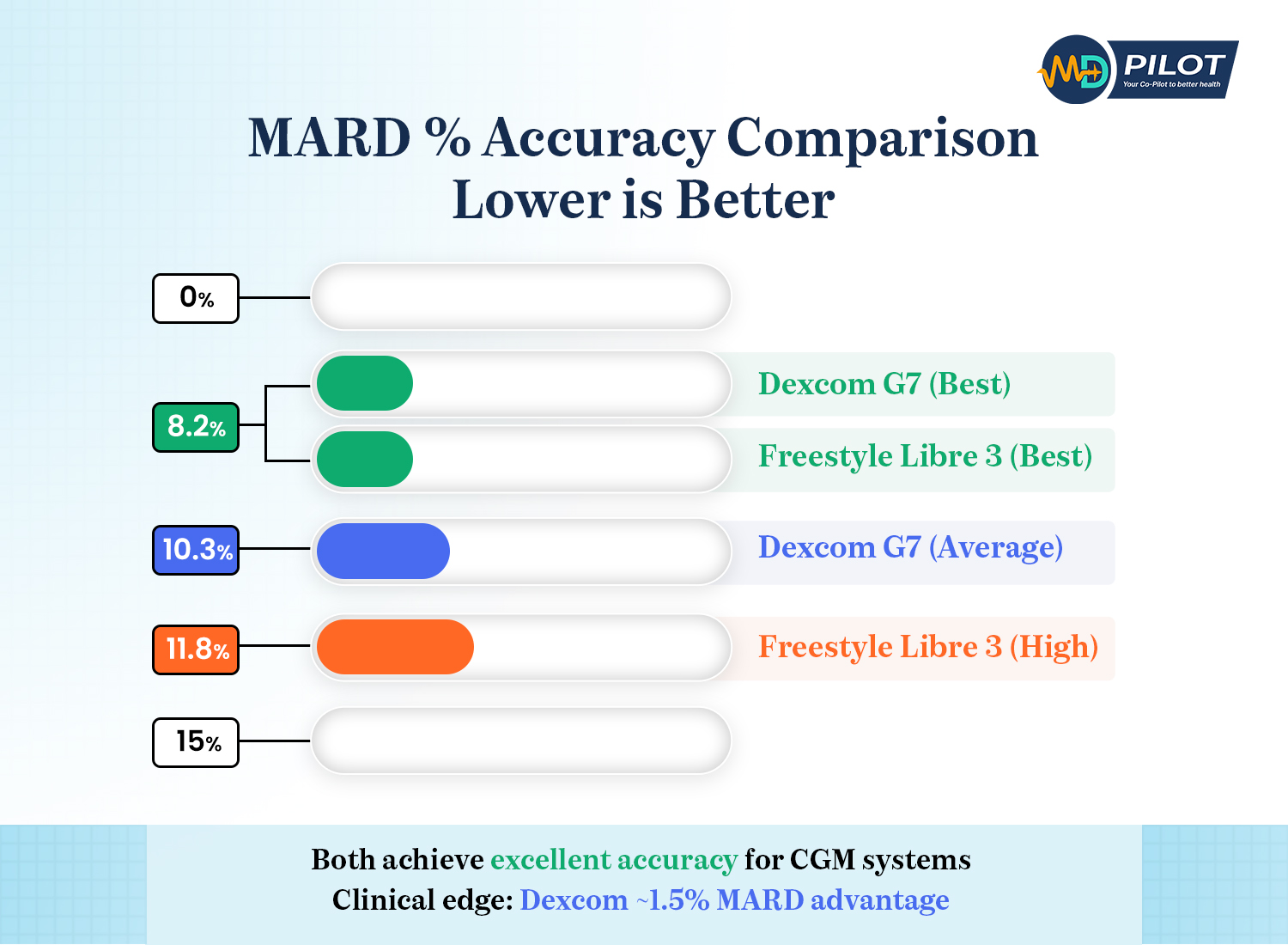
After examining all the clinical data, real-world studies, and user experiences, here’s the bottom line: both Freestyle Libre 3 and Dexcom G7 are excellent continuous glucose monitors with impressive accuracy that can transform your diabetes management. Dexcom G7 holds a slight edge in overall accuracy metrics, while Freestyle Libre 3 offers a more compact sensor design and has traditionally provided longer wear duration (though Dexcom’s new 15-day sensor levels that playing field).
The truth is, choosing between them often comes down to factors beyond pure accuracy numbers. Consider what matters most to you: Do you value the predictive alerts and extensive data sharing capabilities that Dexcom offers? Or do you prefer Freestyle Libre’s compact sensor and streamlined approach? Does your insurance cover one system better than the other? Do you need integration with an insulin pump system?
Both devices have achieved non-adjunctive FDA clearance, meaning they’re accurate enough to make diabetes treatment decisions without confirmatory fingersticks. Both offer factory calibration, real-time alerts, and smartphone connectivity. Both provide the accuracy needed to improve your time in range and overall glycemic control.
If you’re still unsure which system might work better for you, talk with your healthcare provider about trying one system for a few months. Many people find that living with a CGM for a while reveals preferences they didn’t anticipate from reading specifications alone. Some insurance plans and manufacturers offer trial programs that let you experience the technology before committing long-term.
Whichever system you choose, you’ll be joining millions of people with diabetes who have discovered that continuous glucose monitoring—with its minute-by-minute insights, trend arrows, and predictive alerts—makes diabetes management feel less like guesswork and more like informed decision-making. And that might be the most important accuracy improvement of all.