Table Of Contents
-
Key Highlights
- What Are Continuous Glucose Monitors and How Do They Work?
- FDA-Approved OTC Continuous Glucose Monitors in 2025
- Who Can Use OTC Continuous Glucose Monitors?
- Clinical Evidence for CGM Use Without Prescription
- Safety, Device Recalls, and Data Privacy Concerns
- How to Buy and Use OTC Continuous Glucose Monitors
- Insurance and Cost Considerations for OTC CGMs
-
Frequently Asked Questions
-
Conclusion
-
References
If you’ve been wondering whether you can buy a continuous glucose monitor without a prescription, 2025 brings exciting news. The FDA’s clearance of the first over-the-counter continuous glucose monitor has revolutionized access to glucose monitoring technology. No longer limited to people with diabetes on intensive insulin therapy, CGM devices are now available to millions more Americans.
This comprehensive guide covers everything you need to know about continuous glucose monitors without prescription—from FDA-cleared OTC devices and clinical effectiveness to safety concerns, insurance coverage, and practical purchasing advice. Whether you have type 2 diabetes, gestational diabetes, or you’re simply interested in wellness monitoring, you’ll discover how these devices work and whether they’re right for you.
Key Highlights
- In March 2024, the FDA cleared the first OTC continuous glucose monitor (Dexcom Stelo), marking a significant milestone that expanded access to CGM technology without requiring a prescription for adults 18+ not on insulin.
- CGMs provide real-time glucose data every few minutes, replacing traditional finger-stick testing and improving diabetes management awareness through metrics like time in range.
- Clinical evidence shows modest benefits for CGM use in adults with type 2 diabetes on insulin (HbA1c reduction of ~0.27%), with emerging evidence in non-insulin users and wellness applications.
- CGM use in people without diabetes is growing for wellness purposes, but expert clinicians show poor consensus on interpreting these data and determining appropriate follow-up care.
- Safety profile is generally favorable with mild skin irritation being the most common side effect, though recent FDA recalls highlight the importance of monitoring device safety alerts.
- Insurance coverage is expanding but complex—most policies still require prescriptions and insulin use, though OTC availability may gradually change payer policies and improve access.
- Cost-effectiveness data supports CGM use in specific populations, with devices like FreeStyle Libre showing economic benefits over traditional monitoring in basal insulin users.
What Are Continuous Glucose Monitors and How Do They Work?
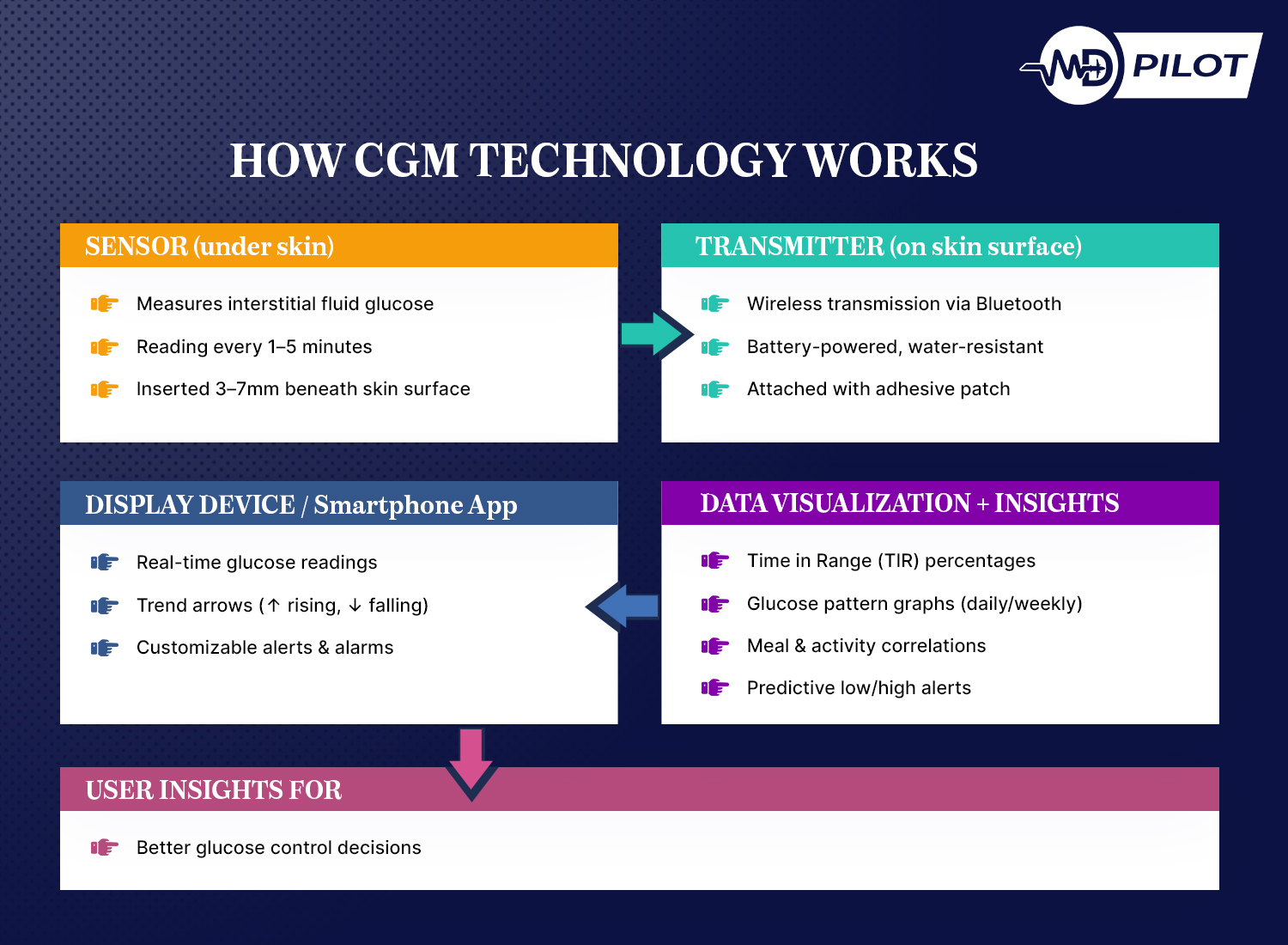
A continuous glucose monitor is a wearable medical device that tracks your blood sugar levels automatically throughout the day and night. Unlike traditional blood glucose meters that require finger-stick tests multiple times daily, CGMs use a tiny sensor inserted just under your skin to measure glucose in your interstitial fluid. This sensor transmits readings to a display device or smartphone app every few minutes, giving you a complete picture of your glucose patterns.
Understanding CGM Technology Types
According to the Washington State Health Technology Assessment, there are two main types of CGM devices available today:
- Real-Time CGM (rtCGM)—continuously transmits glucose readings to your display device and can alert you when levels go too high or too low. Examples include Dexcom G7 and the new OTC Dexcom Stelo.
- Intermittently Scanned CGM (isCGM)—stores glucose data that you access by scanning the sensor with a reader device or phone. The FreeStyle Libre series is the most popular isCGM system.
Key Glucose Metrics You Should Know
Modern CGM systems track several important metrics beyond just your current glucose level. Time in range (TIR) measures the percentage of time your glucose stays between 70-180 mg/dL—research shows that higher TIR correlates with better long-term health outcomes. The glucose management indicator (GMI) estimates what your HbA1c level would be based on your average CGM readings over time.
Most CGM sensors last 10-14 days before requiring replacement, though some newer models extend wear time. Many devices no longer require fingerstick calibration, making them truly maintenance-free once applied. The evolution from self-monitoring blood glucose (SMBG) to continuous monitoring represents a major leap forward in diabetes technology—you get hundreds of data points daily instead of just a handful of snapshots.
FDA-Approved OTC Continuous Glucose Monitors in 2025
The landscape of OTC continuous glucose monitors changed dramatically when the FDA cleared the first prescription-free CGM in 2024. Here’s what you need to know about the devices currently available without a prescription.
Dexcom Stelo: The First OTC Real-Time CGM
In March 2024, the FDA cleared Dexcom Stelo as the first over-the-counter rtCGM device. This groundbreaking clearance means adults 18 and older who don’t use insulin can purchase this device without a prescription. Stelo provides continuous glucose monitoring with real-time alerts, making it ideal for people with type 2 diabetes managing their condition through diet, exercise, or non-insulin medications.
The Stelo system features a small wearable sensor that lasts up to 15 days according to manufacturer specifications and pairs directly with your smartphone. Unlike prescription CGMs, Stelo is specifically designed for people who aren’t at risk of severe hypoglycemia, which is why it’s cleared for non-insulin users. You can buy it online or at select pharmacies without any healthcare provider involvement.
Other Available CGM Options
While Stelo is currently the only true OTC CGM, several other devices have expanded accessibility:
- FreeStyle Libre 2 and Libre 3—still require prescriptions in the U.S., but Abbott has indicated interest in pursuing OTC clearance. These isCGM systems are cleared for ages 4 and up and work with or without insulin therapy.
- Dexcom G6 and G7—prescription-only rtCGM systems with more advanced features like customizable alerts and integration with insulin pumps. The G7 offers the smallest, most discreet sensor available.
Who Can Use OTC Continuous Glucose Monitors?
Who Can Use OTC Continuous Glucose Monitors?
According to recent reviews on CGM advances, OTC devices are specifically intended for:
- Adults with type 2 diabetes not using insulin
- People with prediabetes interested in lifestyle management
- Health-conscious individuals tracking glucose for wellness purposes
- Those with gestational diabetes not requiring insulin therapy
It’s important to note that OTC CGMs are NOT appropriate for people using insulin, children under 18, or anyone at risk of severe hypoglycemia. These populations still need prescription devices with more sophisticated alert systems and clinical oversight.
Clinical Evidence for CGM Use Without Prescription
Before investing in a continuous glucose monitor without prescription, you’re probably wondering: does the science actually support their use? Let’s look at what rigorous clinical research tells us.
What the 2025 Washington State Study Found
The comprehensive Washington State Health Technology Assessment published in 2025 reviewed all available evidence on CGM effectiveness. For adults with type 2 diabetes using basal insulin, CGMs showed moderate certainty evidence of reducing HbA1c by approximately 0.27% compared to traditional monitoring. While this might seem small, even modest HbA1c reductions can meaningfully reduce long-term complications.
However, here’s the catch: for people with type 2 diabetes NOT using insulin—the exact population that OTC devices target—the evidence is much thinner. The assessment found insufficient evidence from randomized controlled trials in this population. Most existing studies focused on insulin users, leaving a significant knowledge gap for the emerging OTC market.
CGM Use in Non-Diabetic Populations
One of the most intriguing developments is the growing use of CGMs by people without diabetes for wellness and metabolic health tracking. A fascinating expert clinician survey on CGM interpretation in non-diabetics revealed significant challenges in this area.
When presented with CGM reports from individuals without diabetes, expert clinicians showed poor consensus on interpretation and follow-up recommendations. Most experts suggested clinical follow-up if CGM data showed more than 2% of time with glucose above 180 mg/dL, but there was wide disagreement on what specific tests to order or when to diagnose prediabetes or diabetes based on CGM patterns alone.
This lack of standardized interpretation guidelines represents a real challenge for OTC CGM users. Without clear guidance on what your numbers mean or when to seek medical advice, you could either overreact to normal glucose variability or miss genuine warning signs of metabolic dysfunction.
Cost-Effectiveness Evidence
From an economic perspective, studies show that CGMs can be cost-effective in specific populations. Research published in 2024 found that the FreeStyle Libre system was dominant over traditional monitoring (meaning both more effective AND less expensive) over a 10-year horizon in Medicaid populations with type 2 diabetes on basal insulin. Cost savings came primarily from preventing costly diabetes complications like kidney disease and cardiovascular events.
The bottom line? Clinical evidence strongly supports CGM use in insulin-using populations, shows emerging benefits in non-insulin users, but lacks clear guidelines for wellness monitoring in people without diabetes. If you’re considering an OTC CGM, it’s worth discussing interpretation strategies with your healthcare provider, even though a prescription isn’t required.
Safety, Device Recalls, and Data Privacy Concerns
Before purchasing any medical device, you should understand the potential risks. While continuous glucose monitors are generally safe, there are some important safety considerations you need to know about.
Common Side Effects and Adverse Events
According to the Washington State HTA analysis, which reviewed 12 randomized controlled trials, 64 device-related adverse events were reported. The vast majority were mild to moderate skin irritation at the sensor insertion site. Some users develop redness, itching, or mild allergic reactions to the adhesive that keeps the sensor attached to your skin.
Serious adverse events are rare but have occurred. The most concerning risks include:
- Sensor accuracy issues that could lead to inappropriate treatment decisions if you rely solely on CGM readings without confirming with fingerstick tests when symptoms don’t match the numbers
- Skin infections at the insertion site, though these are uncommon with proper hygiene practices
- Over-reliance on technology leading to delayed recognition of dangerous glucose levels if you ignore device alerts or your device malfunctions
FDA Recalls and Safety Alerts You Should Know About
The FDA’s MAUDE database (Manufacturer and User Facility Device Experience) shows 649 CGM-related reports between 2019 and 2024, with 5 open device recalls including 4 Class I recalls (the most serious type). Recent recalls have included:
- Battery fire risk in certain Dexcom receivers (though not affecting sensor accuracy or smartphone apps)
- Sensor inaccuracies in specific manufacturing lots that could display falsely high or low readings
- Adhesive failures causing premature sensor detachment
If you’re using a CGM, it’s crucial to register your device with the manufacturer so you receive notifications about recalls affecting your specific lot numbers. Never ignore accuracy alerts from your device, and always confirm with a fingerstick test if your CGM reading doesn’t match how you feel.
Data Security and Privacy Considerations
Modern CGM systems connect to smartphone apps and cloud servers, raising important data privacy concerns. Your glucose data could potentially reveal sensitive health information, daily routines, eating patterns, and activity levels. Before using any CGM system, review:
- What data the manufacturer collects and how it’s encrypted
- Whether your data is shared with third parties or used for research
- How to delete your data if you stop using the device
- Privacy implications if you share data with family members through device apps
Experts from University of Colorado Anschutz point out that expanded OTC access to CGMs without clinical oversight means users bear more responsibility for understanding device limitations and knowing when to seek medical advice for concerning readings.
How to Buy and Use OTC Continuous Glucose Monitors
Ready to purchase a continuous glucose monitor without a prescription? Here’s your practical step-by-step guide to buying and using OTC CGM devices effectively.
Where and How to Purchase OTC CGMs
As of 2025, you have several purchasing options for OTC continuous glucose monitors:
- Directly from manufacturers—Dexcom sells Stelo through their website (dexcom.com/stelo) with direct shipping to your home. This often provides the best pricing for subscription plans.
- Online pharmacies—Major retailers like CVS, Walgreens, and Amazon are beginning to stock OTC CGMs in their online stores.
- Local pharmacies—Check with your neighborhood pharmacy, as availability is expanding rapidly. Call ahead to confirm they stock the specific device you want.
Pricing for Dexcom Stelo typically runs around $90-100 per month (check manufacturer websites for current pricing), which includes two 15-day sensors. This is significantly less expensive than prescription CGMs without insurance, which can cost $200-400 monthly.
Setting Up Your OTC CGM Device
Setting up most OTC CGM systems is straightforward:
- Download the companion app to your smartphone and create an account
- Clean and prepare the application site (usually the back of your upper arm)
- Apply the sensor using the included applicator—most systems use a spring-loaded device that inserts the tiny sensor filament quickly
- Wait for sensor warm-up (usually 30-60 minutes) before readings begin
- Follow the app tutorial to understand your glucose data displays and alerts
Interpreting Your CGM Data Without a Doctor
This is where things get tricky. As the expert interpretation study showed, even experienced clinicians struggle with standardized CGM interpretation guidelines. Here’s what you should focus on:
- Time in range (TIR)—aim for more than 70% of readings between 70-180 mg/dL. Higher percentages indicate better glucose control.
- Time above range—if you’re spending more than 5% of time above 250 mg/dL or 25% above 180 mg/dL, this warrants medical evaluation.
- Time below range—any time spent below 70 mg/dL requires attention, as this indicates hypoglycemia.
- Glucose variability—large swings in glucose levels (even if they stay “in range”) may indicate metabolic stress or dietary issues worth discussing with a provider.
When to Seek Medical Advice
Just because you can buy a CGM without a prescription doesn’t mean you should manage everything independently. Seek medical evaluation if you notice:
- Persistent high glucose readings (>180 mg/dL) despite lifestyle changes
- Any episodes of low glucose (<70 mg/dL), especially if you’re not taking diabetes medications
- Unexplained symptoms that don’t match your CGM readings
- Questions about whether your patterns suggest prediabetes or diabetes
According to pharmacy practice perspectives, pharmacists can be an excellent resource for OTC CGM users, providing counseling on device use, basic interpretation, and guidance on when to consult a physician.
Insurance and Cost Considerations for OTC CGMs
One of the biggest questions people have about continuous glucose monitors without prescription is whether insurance will cover them and how costs compare to prescription devices.
Current Insurance Coverage Landscape
Here’s the complicated reality: most insurance plans, including Medicare, still require a prescription for CGM coverage. The Medicare 2023 policy update expanded coverage to include some non-insulin users with problematic hypoglycemia, but you still need a doctor’s order and documentation.
Key coverage requirements typically include:
- Prescription requirement—even for devices available OTC, insurance usually won’t reimburse without a prescription
- Diabetes diagnosis—you must have a qualifying diagnosis (type 1, type 2, or gestational diabetes)
- Treatment criteria—many plans still limit coverage to insulin users or those with documented hypoglycemia issues
- Prior authorization—insurers may require documentation of failed attempts with traditional monitoring before approving CGM coverage
According to the Center for Health Care Strategies analysis, Medicaid coverage varies dramatically by state, with some states offering comprehensive CGM access while others maintain very restrictive policies creating significant access disparities.
Out-of-Pocket Costs: OTC vs Prescription
If you’re paying out-of-pocket, OTC CGMs offer significant savings:
- OTC devices (Dexcom Stelo)—typically around $90-100/month (check manufacturer websites for current pricing)
- Prescription devices without insurance—$200-400/month depending on the system
- Prescription devices with insurance—typically $50-100/month copay, though this varies widely by plan
For people without insurance or with high-deductible plans, OTC options can provide substantial savings. However, if you have good insurance coverage, getting a prescription for a more advanced device might actually cost less than buying OTC.
Cost-Effectiveness from a Healthcare System Perspective
Research shows that CGMs can be cost-effective by preventing expensive diabetes complications. A 2024 microsimulation study found that FreeStyle Libre was not just cost-effective but actually cost-saving over 10 years in Medicaid populations with type 2 diabetes on basal insulin. The devices paid for themselves through reduced hospitalizations, emergency visits, and complication management.
The American Diabetes Association advocates for expanded insurance coverage of CGMs, arguing that improved access could reduce healthcare disparities and improve population-level diabetes outcomes. As OTC devices become more established, payer policies will likely evolve, though the timeline for broad coverage remains uncertain.
Strategies to Reduce CGM Costs
If cost is a barrier, consider these options:
- Manufacturer savings programs—Dexcom and Abbott offer discount programs for uninsured or underinsured individuals
- Flexible spending accounts (FSAs)—CGMs qualify as eligible medical expenses even without a prescription
- Patient assistance programs—non-profit organizations sometimes provide free or subsidized CGMs for qualifying individuals
- Prescription option—if you qualify, getting a prescription might provide better insurance coverage than buying OTC
Frequently Asked Questions
Yes! In March 2024, the FDA cleared Dexcom Stelo as the first over-the-counter continuous glucose monitor, marking a significant milestone in diabetes technology accessibility. Adults 18 and older who don’t use insulin can purchase this device without a prescription online, through pharmacies, or directly from the manufacturer. However, if you use insulin or are under 18, you still need a prescription for appropriate CGM devices with more advanced safety features.
Focus on key metrics like time in range (aim for >70% between 70-180 mg/dL), time above range (<25% above 180 mg/dL), and time below range (<5% below 70 mg/dL). Look for patterns rather than individual readings—note how different foods, activities, stress, and sleep affect your glucose. Most CGM apps provide helpful visualizations and summaries. However, interpretation can be challenging, and research shows even experts disagree on guidelines, especially for non-diabetics. Consider discussing your CGM reports with a pharmacist or healthcare provider periodically, even though you didn’t need a prescription to purchase the device.
Conclusion
The availability of continuous glucose monitors without prescription represents a significant shift in diabetes technology access. With the FDA’s clearance of OTC devices like Dexcom Stelo, millions of Americans can now access real-time glucose monitoring without navigating the prescription process. This expanded access has the potential to improve diabetes management, increase metabolic awareness, and reduce healthcare disparities.
However, OTC access also comes with new responsibilities. Without automatic clinical oversight, you need to understand device limitations, know when your glucose patterns warrant medical evaluation, and navigate the complex landscape of insurance coverage and out-of-pocket costs. The clinical evidence supporting CGM use is strongest for insulin users, with emerging but less robust data for non-insulin users and wellness applications.
If you’re considering a continuous glucose monitor, start by determining whether you’re a good candidate for OTC devices (adults 18+ not using insulin) or whether you need a prescription device. Compare the costs of cash-pay OTC options versus insured prescription devices to find the most economical approach. Most importantly, view your CGM as a tool that works best alongside professional medical guidance, not as a replacement for it.
The future of continuous glucose monitoring is moving toward greater accessibility, improved accuracy, and longer wear times. As technology advances and insurance coverage evolves, more people will benefit from this powerful diabetes management tool. Whether you have diabetes or you’re exploring metabolic health, OTC CGMs offer an unprecedented window into how your body processes glucose—just make sure you’re prepared to interpret what you see through that window.
References
- Washington State Health Technology Assessment. (2025). Continuous Glucose Monitoring (CGM) Draft Report. Available at: https://www.hca.wa.gov/assets/program/cgm-draft-report-2025.pdf
- Petrak, R. M., et al. (2025). Expert Clinical Interpretation of Continuous Glucose Monitor Reports From Individuals Without Diabetes. BMC Endocrine Disorders. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC11822776/
- Battelino, T., et al. (2025). Continuous Glucose Monitoring Advances and Impact of FDA Over-the-Counter Approval. Health Science Reports. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC12007415/
- Miller, E., & Kerr, M. (2025). Over-the-Counter and Prescription Continuous Glucose Monitors: A Pharmacy Practice Review. PubMed. Available at: https://pubmed.ncbi.nlm.nih.gov/40105027/
- U.S. Food and Drug Administration. (2024). FDA Clears First Over-the-Counter Continuous Glucose Monitor. Press announcement. Available at: https://www.fda.gov/news-events/press-announcements/fda-clears-first-over-counter-continuous-glucose-monitor
- Centers for Medicare & Medicaid Services. (2023). Local Coverage Determination for Continuous Glucose Monitors. LCD ID 33822. Available at: https://www.cms.gov/medicare-coverage-database/view/lcd.aspx?lcdid=33822
- Center for Health Care Strategies. (2024). Continuous Glucose Monitor Access for Medicaid Beneficiaries Living with Diabetes: State-by-State Coverage. Available at: https://www.chcs.org/resource/continuous-glucose-monitor-access-for-medicaid-beneficiaries-living-with-diabetes-state-by-state-coverage/
- American Diabetes Association. (2025). Advocacy for CGM Access and Coverage. Available at: https://diabetes.org/advocacy/cgm-continuous-glucose-monitors
- University of Colorado Anschutz Medical Campus. (2024). Continuous Glucose Monitors for All? Opinions Remain Mixed. Available at: https://news.cuanschutz.edu/medicine/continuous-glucose-monitors-for-all-opinions-remain-mixed
- Frank, J. W., et al. (2024). Real-World Cost-Effectiveness of Continuous Glucose Monitoring in Type 2 Diabetes: A Medicaid Microsimulation Analysis. Diabetes Care (manufacturer data).
Disclaimer:
The information provided on MD-Pilot is for educational and informational purposes only. It is not intended as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified healthcare provider with any questions you may have regarding a medical condition. Never disregard professional medical advice or delay in seeking it because of something you have read on this website.
Recomended Articles
View AllWeekly Health Intel
Get evidence-based health tips, latest research, and exclusive guides delivered weekly